Nitric Acid Formula Oxidation Number . using this information we can figure out the oxidation number for the. A net ionic charge can be specified at the. According to rule 4, the oxidation number of hydrogen is \(+1\). N 2 o 5 + h 2 o → hno 3. nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. as an example, consider the compound nitric acid, \(\ce{hno_3}\). nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. enter the formula of a chemical compound to find the oxidation number of each element. nitric acid is obtained by mixing dinitrogen pentoxide (n 2 o 5) and water giving pure hno 3.
from www.laboratorynotes.com
nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. enter the formula of a chemical compound to find the oxidation number of each element. nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. N 2 o 5 + h 2 o → hno 3. using this information we can figure out the oxidation number for the. A net ionic charge can be specified at the. According to rule 4, the oxidation number of hydrogen is \(+1\). as an example, consider the compound nitric acid, \(\ce{hno_3}\). nitric acid is obtained by mixing dinitrogen pentoxide (n 2 o 5) and water giving pure hno 3.
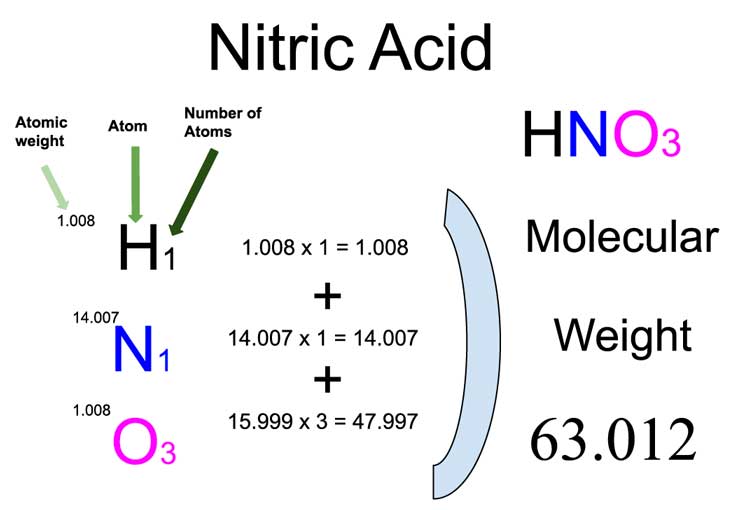
Nitric Acid Molecular Weight Calculation Laboratory Notes
Nitric Acid Formula Oxidation Number nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. According to rule 4, the oxidation number of hydrogen is \(+1\). nitric acid is obtained by mixing dinitrogen pentoxide (n 2 o 5) and water giving pure hno 3. A net ionic charge can be specified at the. nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. enter the formula of a chemical compound to find the oxidation number of each element. nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. as an example, consider the compound nitric acid, \(\ce{hno_3}\). N 2 o 5 + h 2 o → hno 3. using this information we can figure out the oxidation number for the.
From pubs.rsc.org
Electrochemical oxidation of molecular nitrogen to nitric acid Nitric Acid Formula Oxidation Number as an example, consider the compound nitric acid, \(\ce{hno_3}\). nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. using this information we can figure out the oxidation number for the. enter the formula of a chemical. Nitric Acid Formula Oxidation Number.
From www.chemicals.co.uk
What Is Nitric Acid? The Chemistry Blog Nitric Acid Formula Oxidation Number nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. A net ionic charge can be specified at the. N 2 o 5 + h 2 o → hno 3. enter the formula of a chemical compound to find. Nitric Acid Formula Oxidation Number.
From www.youtube.com
How to find the Oxidation Number for N in NO3 (Nitrate ion) YouTube Nitric Acid Formula Oxidation Number N 2 o 5 + h 2 o → hno 3. using this information we can figure out the oxidation number for the. nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. nitric acid is obtained by. Nitric Acid Formula Oxidation Number.
From www.dreamstime.com
Nitric acid molecule stock illustration. Illustration of formula Nitric Acid Formula Oxidation Number enter the formula of a chemical compound to find the oxidation number of each element. According to rule 4, the oxidation number of hydrogen is \(+1\). using this information we can figure out the oxidation number for the. N 2 o 5 + h 2 o → hno 3. nitric acid is a nitrogen oxoacid of formula. Nitric Acid Formula Oxidation Number.
From www.nagwa.com
Question Video Identifying the Oxidation Number of Nitrogen in Nitric Nitric Acid Formula Oxidation Number N 2 o 5 + h 2 o → hno 3. enter the formula of a chemical compound to find the oxidation number of each element. using this information we can figure out the oxidation number for the. nitric acid is obtained by mixing dinitrogen pentoxide (n 2 o 5) and water giving pure hno 3. . Nitric Acid Formula Oxidation Number.
From mavink.com
Nitric Acid Lewis Structure Nitric Acid Formula Oxidation Number using this information we can figure out the oxidation number for the. nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. as an example, consider the compound nitric acid, \(\ce{hno_3}\). According to rule 4, the oxidation number of hydrogen is \(+1\). A net ionic charge can be specified at the. nitric. Nitric Acid Formula Oxidation Number.
From dxoccdzwi.blob.core.windows.net
Nitric Oxide Formula With at Shakira Thornton blog Nitric Acid Formula Oxidation Number N 2 o 5 + h 2 o → hno 3. enter the formula of a chemical compound to find the oxidation number of each element. A net ionic charge can be specified at the. as an example, consider the compound nitric acid, \(\ce{hno_3}\). nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen. Nitric Acid Formula Oxidation Number.
From www.onlinechemistrytutor.net
Oxidation state examples Online Chemistry Tutor Nitric Acid Formula Oxidation Number A net ionic charge can be specified at the. nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. enter the formula of a chemical compound to find the oxidation number of each element. as an example, consider the compound nitric acid, \(\ce{hno_3}\). N 2 o 5 + h 2 o → hno. Nitric Acid Formula Oxidation Number.
From www.youtube.com
NO2+H2O=HNO3+NO Balanced EquationNitrogen dioxide+Water=Nitric acid Nitric Acid Formula Oxidation Number nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. N 2 o 5 + h 2 o → hno 3. enter the formula of a chemical compound to find the oxidation number of each element. as an. Nitric Acid Formula Oxidation Number.
From www.numerade.com
SOLVED Assign oxidation numbers to each of the elements in the Nitric Acid Formula Oxidation Number nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. using this information we can figure out the oxidation number for the. as an example, consider the compound nitric acid, \(\ce{hno_3}\). nitric acid is obtained by mixing. Nitric Acid Formula Oxidation Number.
From www.chemistryscl.com
Preparation and Uses of Nitrous Acid Nitric(iii) acid HNO2 Nitric Acid Formula Oxidation Number nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. enter the formula of a chemical compound to find the oxidation number of each element. as an example, consider the compound nitric acid, \(\ce{hno_3}\). A net ionic charge can be specified at the. According to rule 4, the oxidation number of hydrogen is. Nitric Acid Formula Oxidation Number.
From www.laboratorynotes.com
Nitric Acid Molecular Weight Calculation Laboratory Notes Nitric Acid Formula Oxidation Number N 2 o 5 + h 2 o → hno 3. nitric acid is obtained by mixing dinitrogen pentoxide (n 2 o 5) and water giving pure hno 3. nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. enter the formula of a chemical compound to find the oxidation number of each. Nitric Acid Formula Oxidation Number.
From www.gbu-taganskij.ru
Nitric Acid Or Hno3 Molecule Skeletal Formula Vector Image, 44 OFF Nitric Acid Formula Oxidation Number nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. nitric acid is obtained by mixing dinitrogen pentoxide (n 2 o 5) and water giving pure hno 3. enter the formula of a chemical compound to find the. Nitric Acid Formula Oxidation Number.
From www.numerade.com
SOLVED What is the oxidation number of nitrogen in nitric acid HNO3?a Nitric Acid Formula Oxidation Number According to rule 4, the oxidation number of hydrogen is \(+1\). A net ionic charge can be specified at the. nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds. Nitric Acid Formula Oxidation Number.
From www.youtube.com
How to Calculate the Formal Charges for HNO3 (Nitric acid) YouTube Nitric Acid Formula Oxidation Number as an example, consider the compound nitric acid, \(\ce{hno_3}\). nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. enter the formula of a chemical compound to find the oxidation number of each element. nitric acid is. Nitric Acid Formula Oxidation Number.
From www.numerade.com
SOLVED 369 of RedoxBalancing Which of the following is the correctly Nitric Acid Formula Oxidation Number N 2 o 5 + h 2 o → hno 3. enter the formula of a chemical compound to find the oxidation number of each element. as an example, consider the compound nitric acid, \(\ce{hno_3}\). nitric acid is obtained by mixing dinitrogen pentoxide (n 2 o 5) and water giving pure hno 3. A net ionic charge. Nitric Acid Formula Oxidation Number.
From www.dreamstime.com
Chemical Formulas of Nitrogen Oxide Nitric Oxide NO, Nitrogen Dioxide Nitric Acid Formula Oxidation Number A net ionic charge can be specified at the. enter the formula of a chemical compound to find the oxidation number of each element. nitric acid decomposes into water, nitrogen dioxide, and oxygen, forming a brownish yellow solution. N 2 o 5 + h 2 o → hno 3. using this information we can figure out the. Nitric Acid Formula Oxidation Number.
From www.vectorstock.com
Nitric oxide molecule skeletal formula Royalty Free Vector Nitric Acid Formula Oxidation Number nitric acid is obtained by mixing dinitrogen pentoxide (n 2 o 5) and water giving pure hno 3. as an example, consider the compound nitric acid, \(\ce{hno_3}\). nitric acid is a nitrogen oxoacid of formula hno3 in which the nitrogen atom is bonded to a hydroxy group and by equivalent bonds to the remaining two oxygen. . Nitric Acid Formula Oxidation Number.